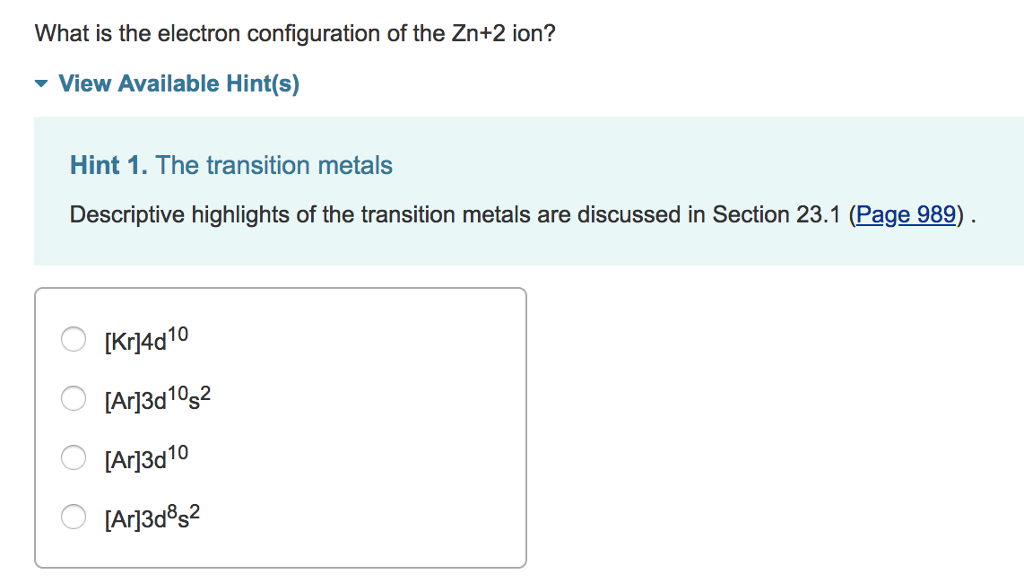
Zn2+ is: a) diamagnetic. b) paramagnetic with one unpaired electron. c) paramagnetic with two unpaired electrons. d) paramagnetic with four unpaired electrons. e) paramagnetic with five unpaired electrons. | Homework.Study.com

Zinc Zn Chemistry Zn2+ compounds oxidation state +2 complexes complex ions chemical reactions GCE AS A2 IB A level inorganic chemistry revision notes
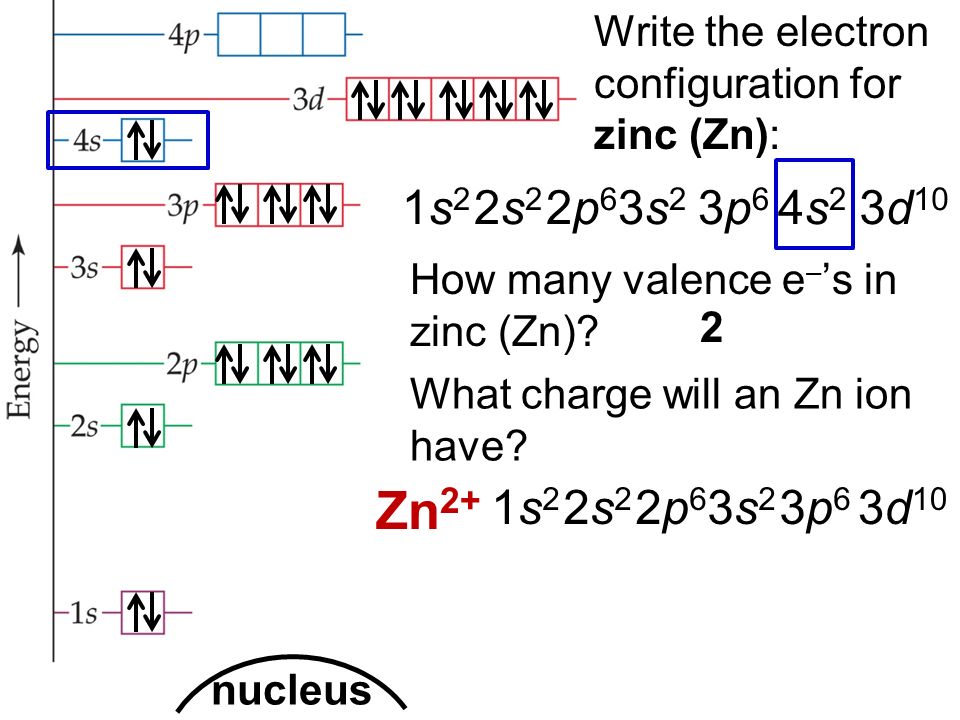
![Draw orbital box diagrams for Fe^2+, Fe^3+, Zn, and Zn^2+. Tell which is paramagnetic. [Paramagnetic means that it has unpaired electrons. This can only be seen with box diagrams.] | Homework.Study.com Draw orbital box diagrams for Fe^2+, Fe^3+, Zn, and Zn^2+. Tell which is paramagnetic. [Paramagnetic means that it has unpaired electrons. This can only be seen with box diagrams.] | Homework.Study.com](https://homework.study.com/cimages/multimages/16/3-3409192489036411813.jpg)
Draw orbital box diagrams for Fe^2+, Fe^3+, Zn, and Zn^2+. Tell which is paramagnetic. [Paramagnetic means that it has unpaired electrons. This can only be seen with box diagrams.] | Homework.Study.com
![SOLVED: Part A Zn2+ ions are represented by the electron configuration [Kr]3d10 [Arj3d8 [Arj4s23d8 [Ar]3d10 [Ar]3d12 Submit Request Answer SOLVED: Part A Zn2+ ions are represented by the electron configuration [Kr]3d10 [Arj3d8 [Arj4s23d8 [Ar]3d10 [Ar]3d12 Submit Request Answer](https://cdn.numerade.com/ask_previews/73aa58ea-e19e-411e-bf16-7e1288f1db02_large.jpg)