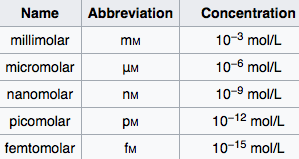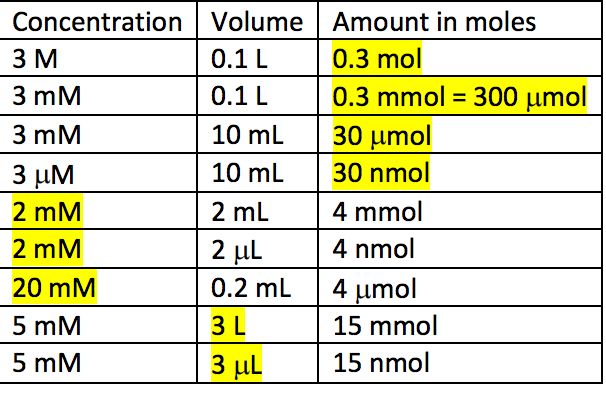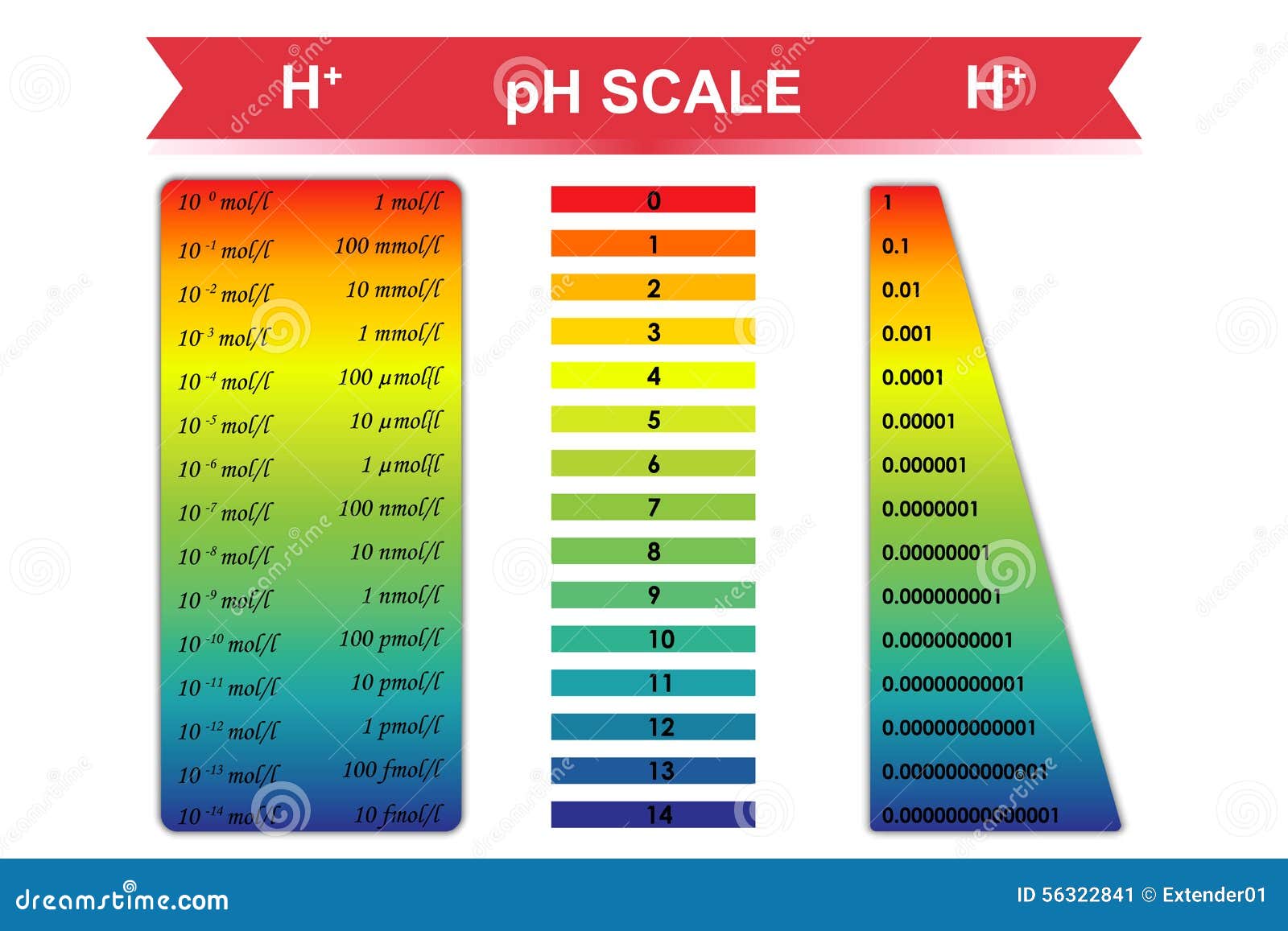
Chemical Mol Stock Illustrations – 22 Chemical Mol Stock Illustrations, Vectors & Clipart - Dreamstime

AMT - Air pollution monitoring: development of ammonia (NH3) dynamic reference gas mixtures at nanomoles per mole levels to improve the lack of traceability of measurements

The pH of the solution is the negative logarithm to the base 10 of its hydrogen ion concentration in .

SOLVED: Ans: Given Values: Amount of p-nitrophenol = 50 nmol The volume of the Tube 500 microliter The formula to calculate molar concentration is as follows: Moles M Liter 50 nmoles S0x10-
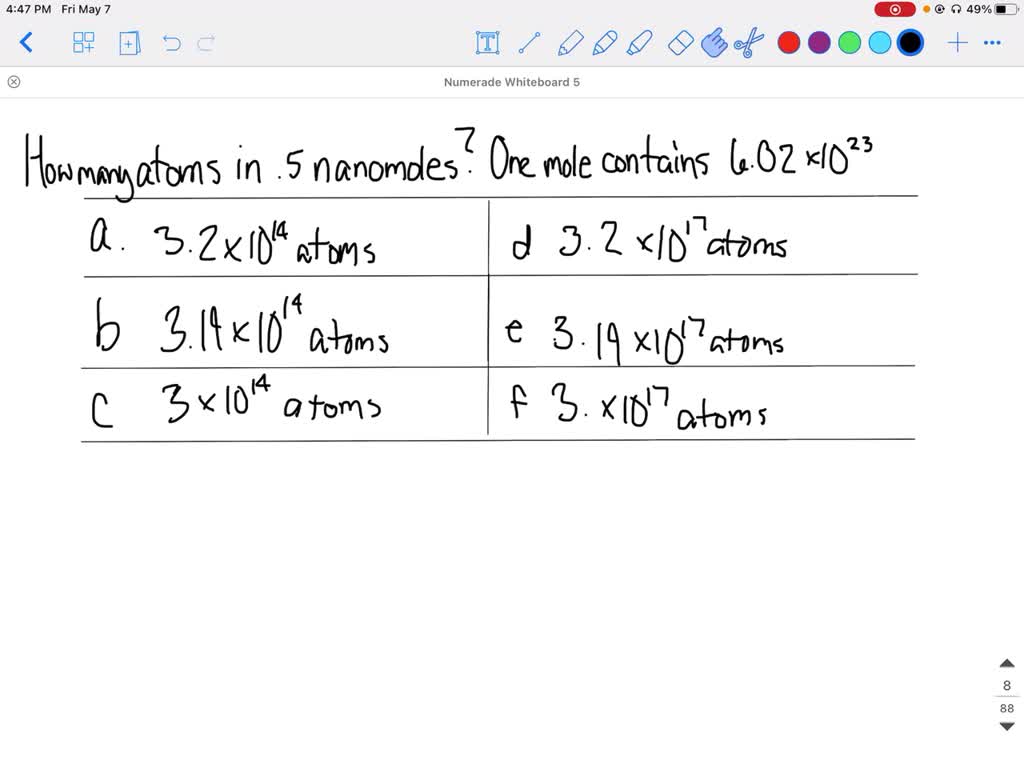
SOLVED:What is the number of carbon atoms in 0.5 nanomoles of carbon? One mole contains 6.02 ·10^23 atoms. a) 3.2 ·10^14 atoms e) 3.19 ·10^17 atoms b) 3.19 ·10^14 atoms f) 3 . ·10^17 atoms c) 3 .+10^14 atoms d) 3.2 ·10^17 atoms