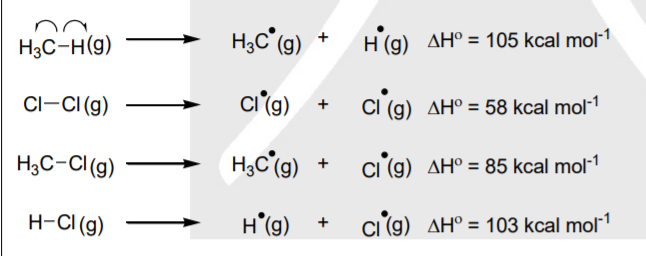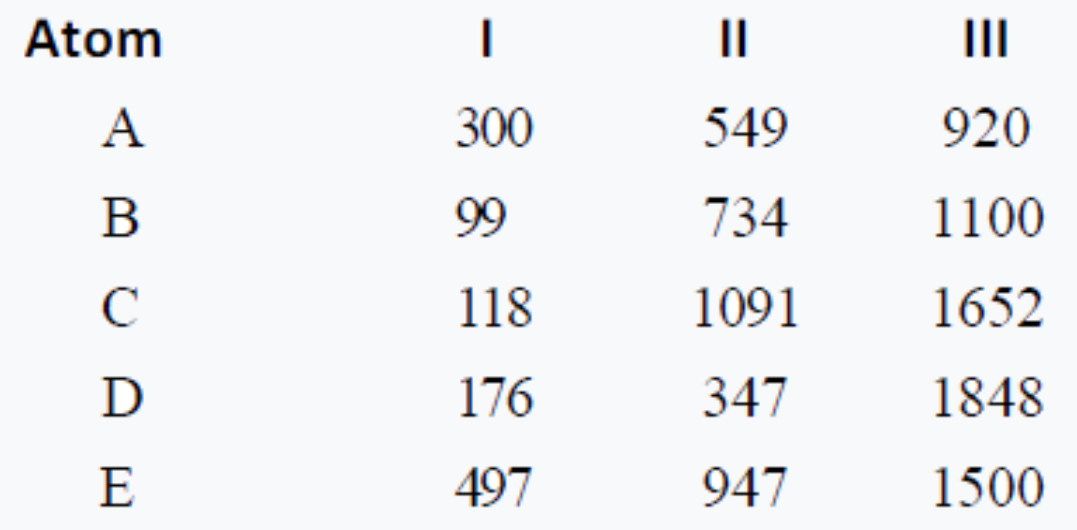
Ionization energies of five elements in kcal/mol are given below: . Which element forms stable unipositive ion ?
24. In the reaction , Y ——–> Z Delta H = + 100 Kcal/mol Z ———> X , Delta H = 80 Kcal/mol Rank the enthalpies of formation of X , Y, and Z in increasing order Options : 1 Y

From the given table answer the following questions :CO(g) CO2(g) H2O(g) H2(g) ( Δ H^∘ f )298 ( - kCal / mole ) - 26.42 - 94.05 - 57.80 ( Δ G^∘

Insertion of Pro-Hyp-Gly provides 2 kcal mol−1 stability but attenuates the specific assembly of ABC heterotrimeric collagen triple helices - Organic & Biomolecular Chemistry (RSC Publishing)
The lattice enthalpy of KI will be, if the enthalpy of (I) Δ_fH^°(KI) = –78.0 kcal mol^ 1 (II) Ionisation energy of K to K+ is 4.0 eV (III) Dissociation energy of
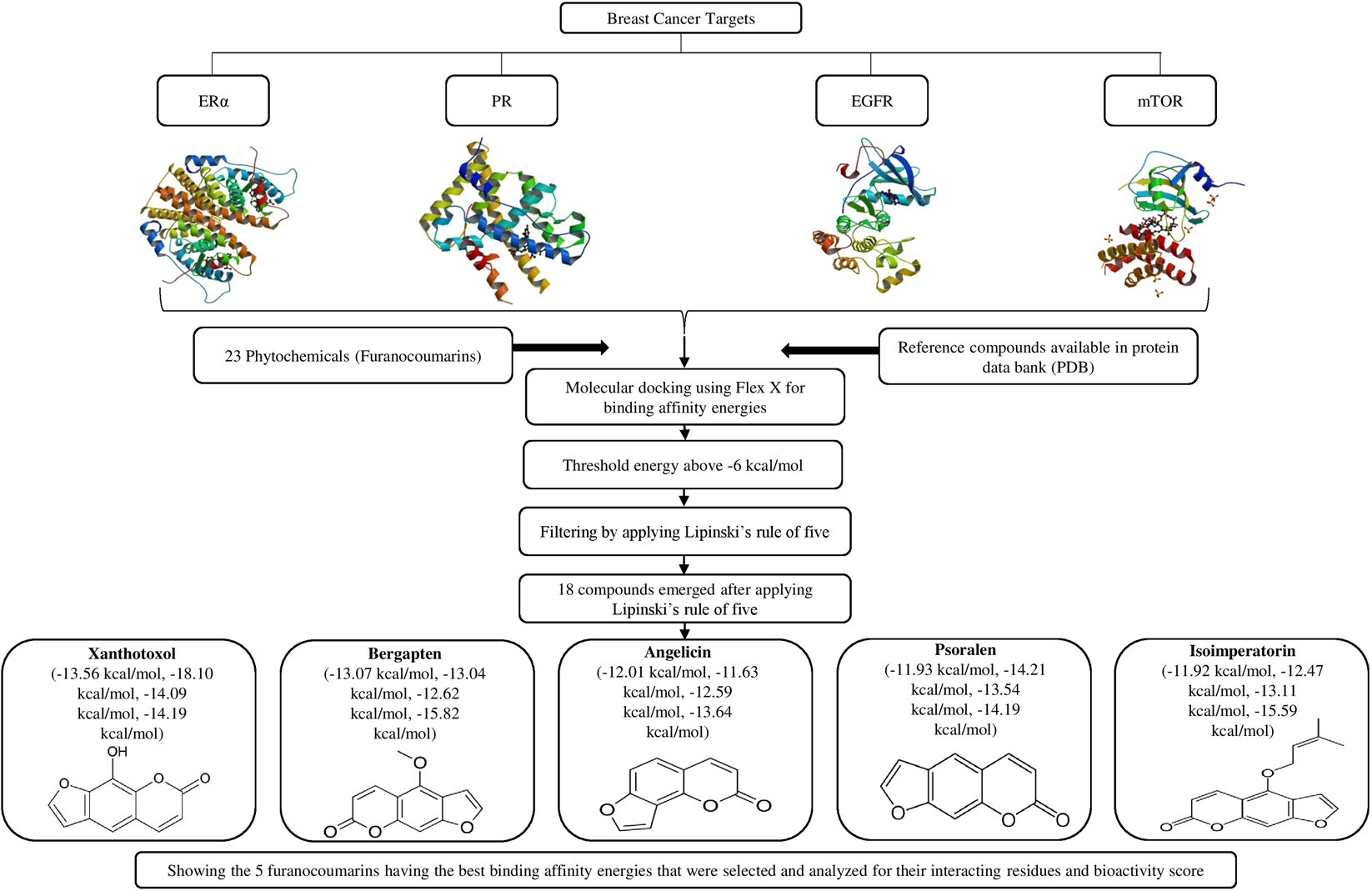
Structure Based Multitargeted Molecular Docking Analysis of Selected Furanocoumarins against Breast Cancer | Scientific Reports
En = –313.6/n2 kcal/mole. If the value of E = –34.84 kcal/mole, to which value does 'n' correspond? (1) 4 (2) 3 (3) 2 (4) 1

Standard heat of formation of KI is - 78.31 kcal mol^-1 . Calculate its lattice energy from following information: IE (K) = 4.3 eV EA (I) = - 73.4 kcal mol^-1 Bond

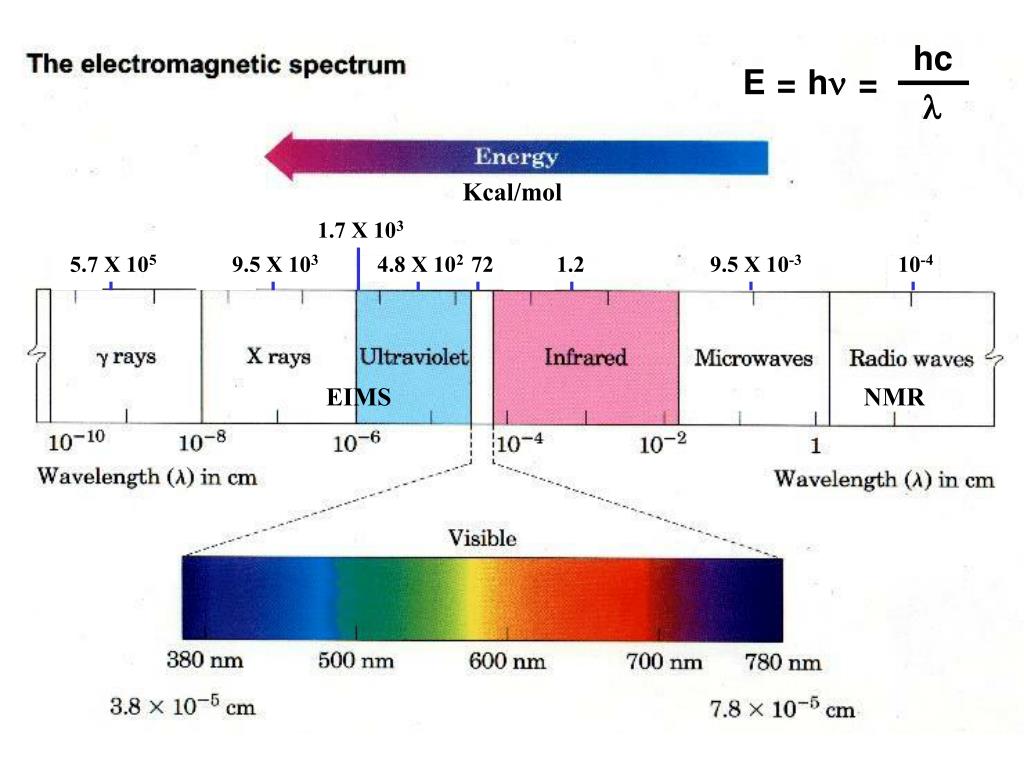
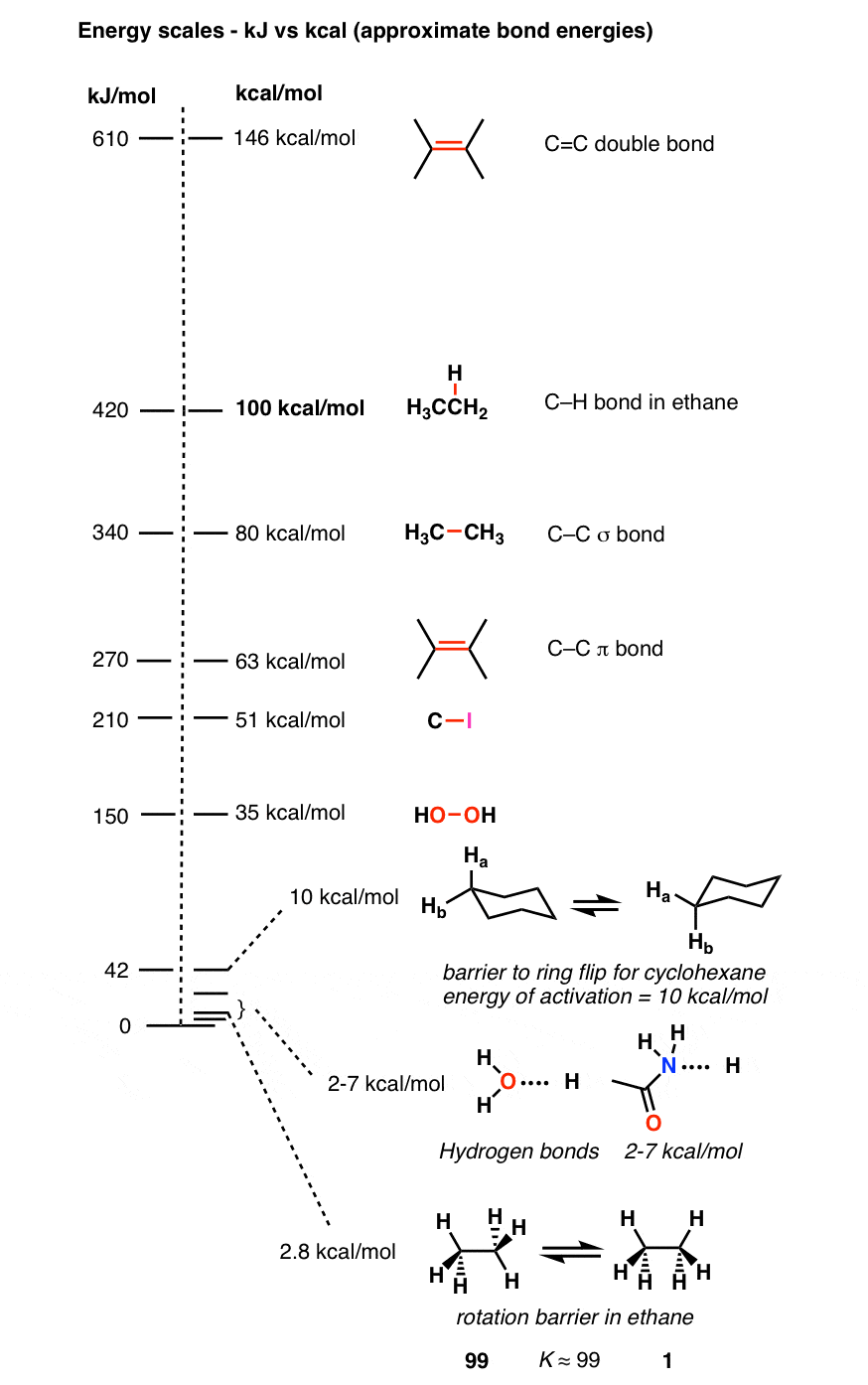
Forces Note on units: Energy kcal/mol(1kcal = kJ). DistanceÅ(1Å = M) At room temperature thermal energy = RT=.59 kcal/mol I. Covalent bonds. - ppt download

Forces Note on units: Energy kcal/mol(1kcal = kJ). DistanceÅ(1Å = M) At room temperature thermal energy = RT=.59 kcal/mol I. Covalent bonds. - ppt download
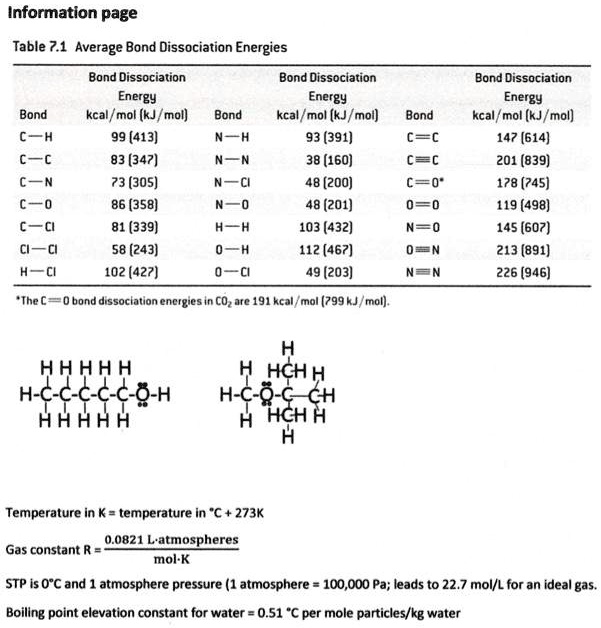
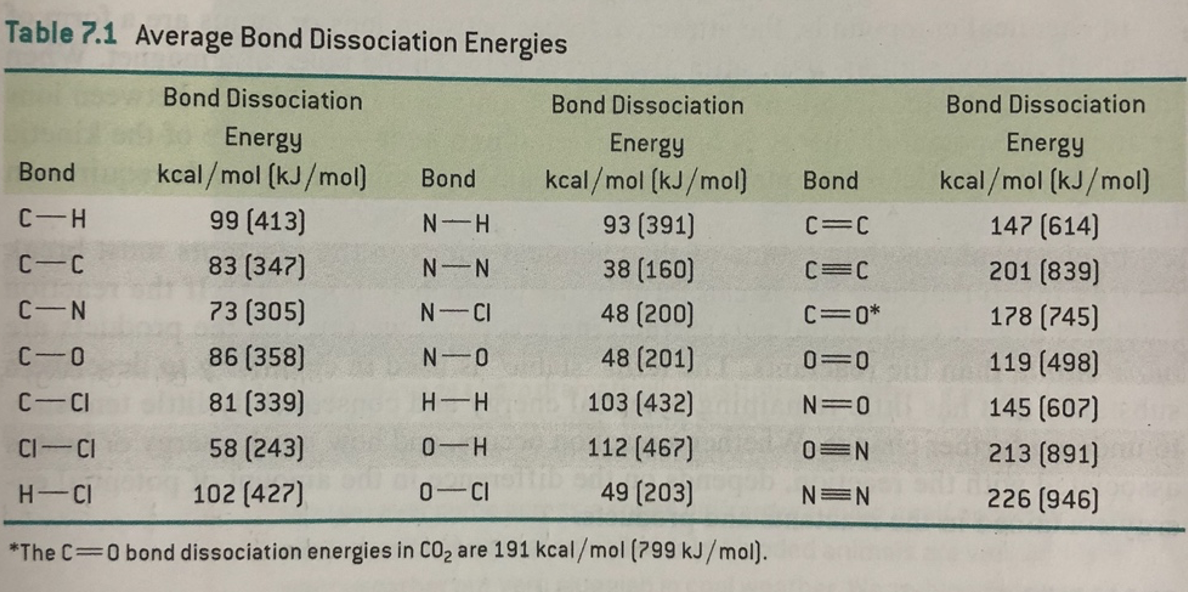
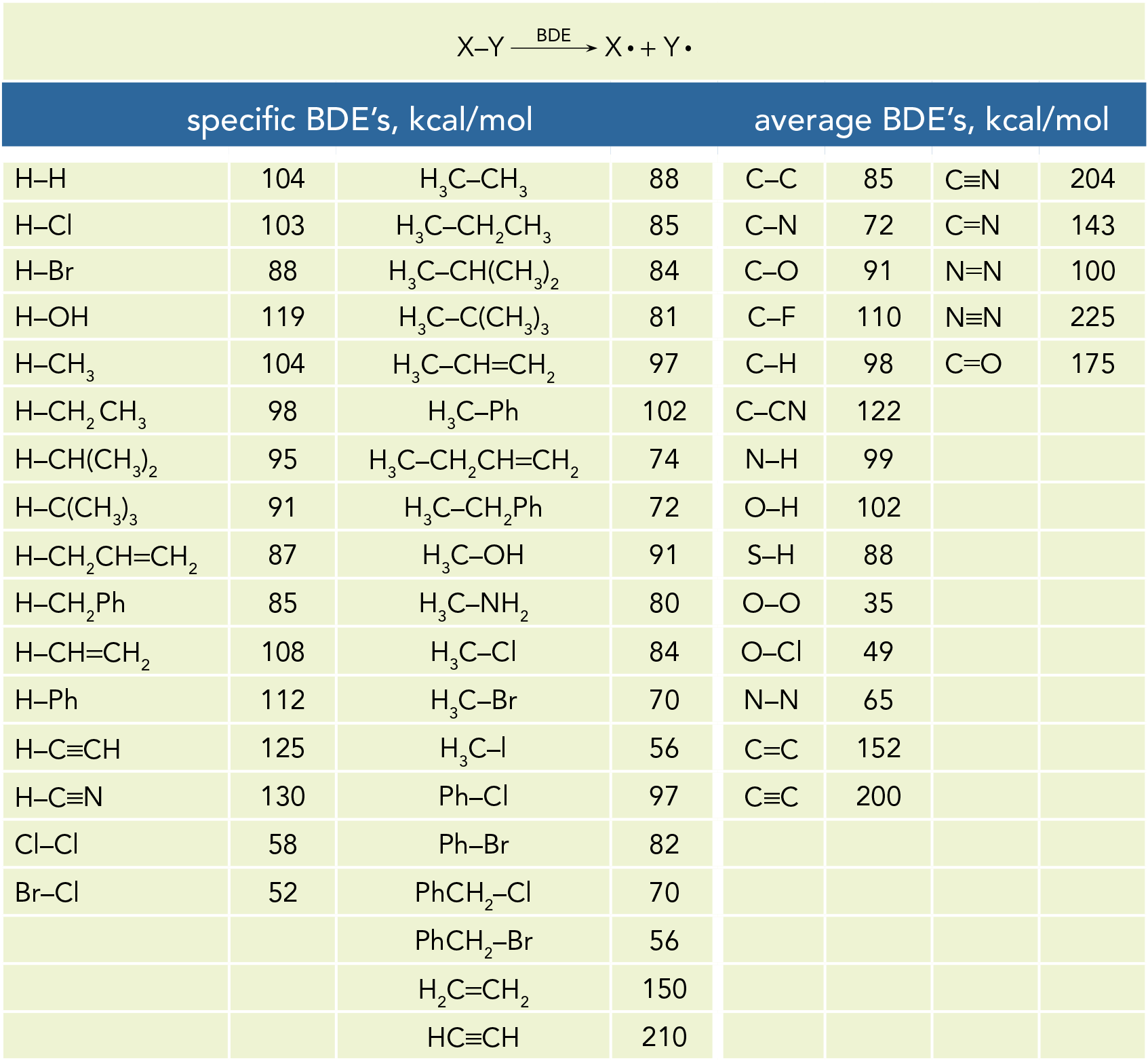
SOLVED: Information page Table ?.1 Average Bond Dissociation Energies Bond Dissociation Bond Dissociation Energy Encrgy Bond kcal / mol (kJ /mol Bond kcal /mol (kJ /moi) CH 99 (413) (391) Cc 83 (

SOLVED: ATP Hydrolysis yields 7.3 Kcal/mol free energy (ΔGo = -7.3 Kcal/mol) and it is coupled with many enzyme reactions that would not otherwise happen. Which of the following reaction(s) can be