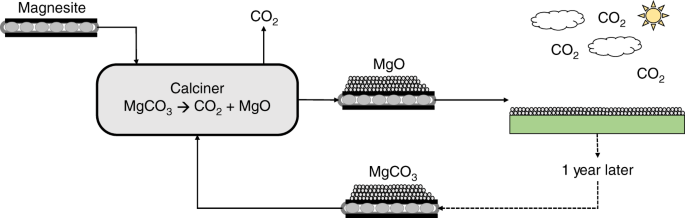
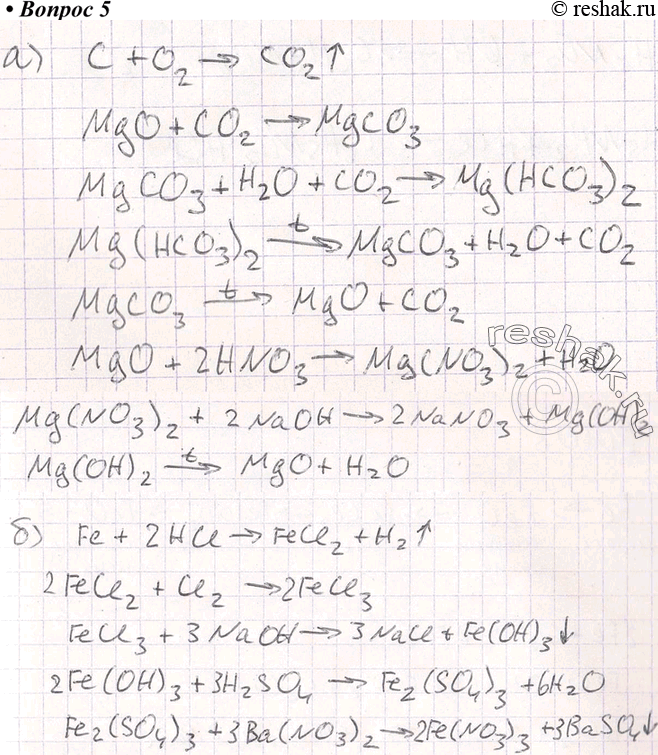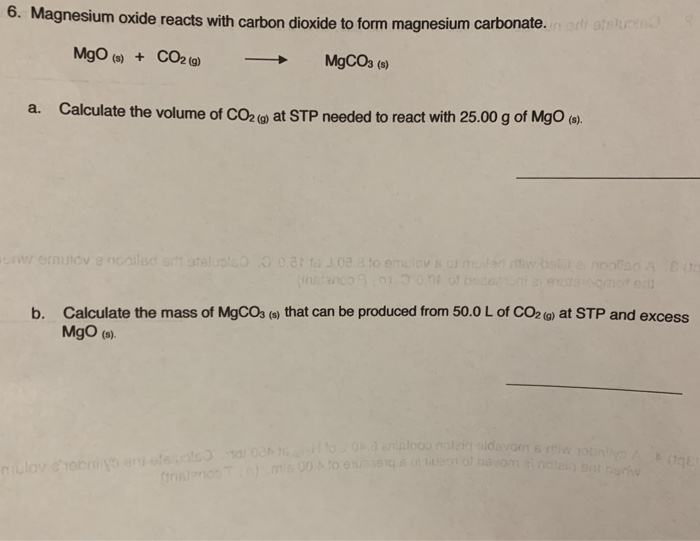Magnesium oxide as an efficient catalyst for CO2 fixation and N-formylation reactions under ambient conditions - ScienceDirect

Mg +CO2 =MgO +C Balanced Equation||Magnesium +Carbon dioxide=Magnesium oxide +Carbon Balanced Equ. - YouTube
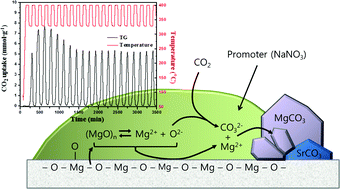
Mechanisms of absorption and desorption of CO2 by molten NaNO3-promoted MgO - Physical Chemistry Chemical Physics (RSC Publishing)
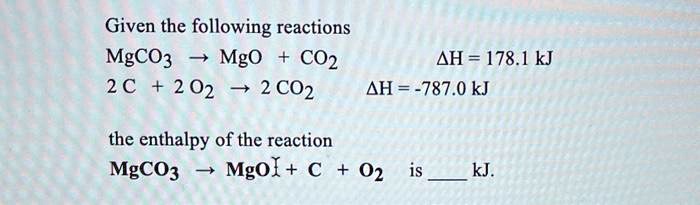
SOLVED: Given the following reactions MgCO3 Mgo CO2 AH = 178.1 kJ 2 C 2 02 2 CO2 AH = -787.0 kJ the enthalpy of the reaction MgCO3 Mgol + € 02 kJ.

CO2-TPD profiles of MgO-450, MgO-550, MgO-650, ZnMg2Ox, ZnMgOx and ZnO. | Download Scientific Diagram

SOLVED: Be sure to answer all parts. Consider the following balanced thermochemical equation for the decomposition of the mineral magnesite: MgCO3(s) → MgO(s) + CO2(g) ΔHrxn = 117.3 kJ (a) Is heat
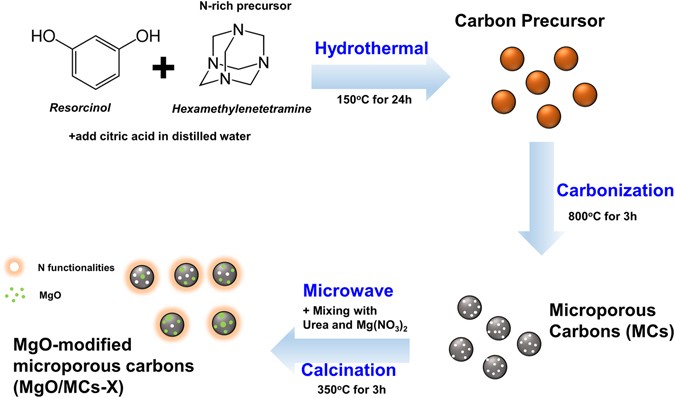
Facile Synthesis of MgO-Modified Carbon Adsorbents with Microwave- Assisted Methods: Effect of MgO Particles and Porosities on CO2 Capture | Scientific Reports
![PDF] Adapting the MgO-CO2 Working Pair for Thermochemical Energy Storage by Doping with Salts: Effect of the (LiK)NO3 Content | Semantic Scholar PDF] Adapting the MgO-CO2 Working Pair for Thermochemical Energy Storage by Doping with Salts: Effect of the (LiK)NO3 Content | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bf56c72e2474994b03ef3acaf38276c7a38f3407/7-Figure5-1.png)
PDF] Adapting the MgO-CO2 Working Pair for Thermochemical Energy Storage by Doping with Salts: Effect of the (LiK)NO3 Content | Semantic Scholar

CO2 Uptake and Cyclic Stability of MgO-Based CO2 Sorbents Promoted with Alkali Metal Nitrates and Their Eutectic Mixtures | ACS Applied Energy Materials
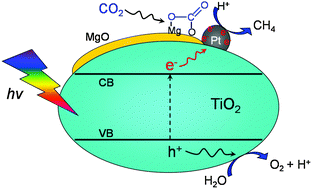
Photocatalytic reduction of CO2 with H2O: significant enhancement of the activity of Pt–TiO2 in CH4 formation by addition of MgO - Chemical Communications (RSC Publishing)

CO2+Mg=MgO+C электронный баланс, окислитель и восстановитель, расставить коэффиценты. - Школьные Знания.com

How to balance MgCO3=MgO+CO2| Chemical equation MgCO3=MgO+CO2|Reaction balance MgCO3=MgO+CO2 - YouTube